CCS Guard™ | Contamination Control is Now Continuous
A compliance platform for sterile pharmaceutical manufacturing environments
Sterile pharmaceutical manufacturing is entering a new phase of regulatory expectation. Annex 1 now requires organisations to implement and continuously demonstrate a structured Contamination Control Strategy across all critical operations. However, most facilities are still operating with fragmented systems, manual processes, and delayed reporting. Environmental monitoring, personnel movement, and operational data are often managed in isolation, making it difficult to maintain a clear, real-time view of risk. This creates a growing disconnect between regulatory requirements and operational reality, where issues are identified too late, trends are missed, and compliance becomes reactive rather than controlled.
The Challenge
Compliance Requirements Have Changed – Most Systems Haven’t
The Solution
CCS Guard provides a unified compliance layer that integrates with existing systems and transforms operational data into structured, real-time compliance intelligence.
Rather than relying on fragmented tools and retrospective reporting, organisations gain continuous visibility across contamination control processes, enabling proactive decision-making and consistent regulatory alignment.
The platform shifts compliance from a manual burden into a live operational framework embedded within day-to-day manufacturing environments.
A pilot customer is already preparing to deploy CCS Guard, demonstrating real-world demand and readiness for implementation.
System Integration
Automated Risk Detection
Continuous Trend Analysis
Audit-Ready Documentation
Platform Overview
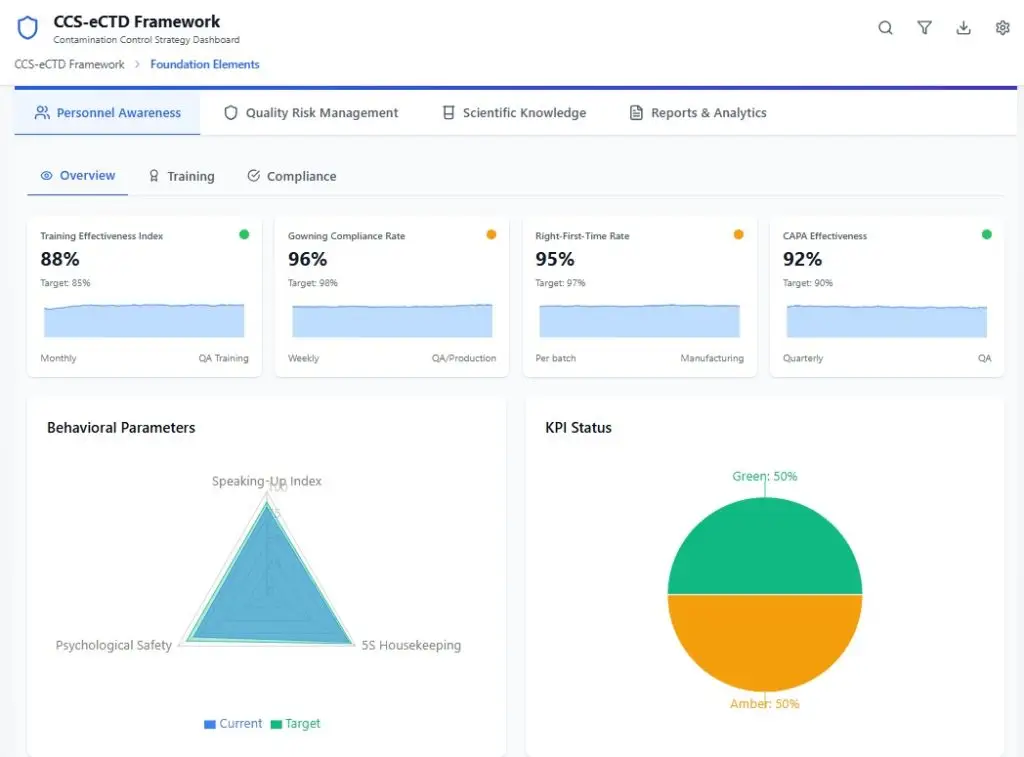
CCS Guard is a compliance platform that integrates with existing manufacturing systems, transforming operational data into real-time regulatory intelligence.
CCS Guard transforms compliance from retrospective reporting into a continuous, operational control system.
Our Core Capabilities
Provision of a 'Single Source of Truth'.
Real-time monitoring of contaminants, environmental conditions, and personnel flow between controlled areas enables proactive identification of hidden risks across staffing practices, training effectiveness, and facility infrastructure.
System Integration
Connects with BMS, EMS, LIMS and QMS systems to create a unified data environment without disrupting existing operations
Data Aggregation
Collects and centralises operational and environmental data from multiple sources into a single structured framework.
Data Structuring
Standardises and maps data into contamination control frameworks aligned with regulatory expectations.
Real-Time Risk Detection
Continuously analyses data to identify deviations, emerging risks, and compliance gaps in real time.
Continuous Compliance Monitoring
Provides ongoing visibility into environmental performance and process stability through trend analysis
Audit Ready Output
Generates structured, regulator-ready documentation automatically, reducing manual effort and ensuring consistent compliance.
From Advanced Prototype to Enterprise Deployment
Current Position
CCS Guard has moved beyond concept and is now positioned for initial enterprise deployment.
The core platform is in place, with a working demonstration environment that reflects real-world compliance conditions. The regulatory framework has been aligned with Annex 1 requirements, and early engagement with industry stakeholders is underway.
A pilot customer is already in place and preparing to use the platform, showing clear demand and a natural transition into real-world deployment.
The focus now is on moving from advanced prototype to controlled rollout, with pilot deployment supporting validation and readiness for scale.
Collaboration & Strategic Engagement
We are engaging with a limited number of organisations to support CCS Guard’s next phase of deployment. This includes pharmaceutical manufacturers, compliance specialists, and integration partners interested in pilot deployment, validation, and early-stage rollout. Engagement is structured through direct, private discussion, with detailed technical and commercial information shared where appropriate.

We are engaging with a limited number of organisations operating within sterile pharmaceutical manufacturing, compliance, or enterprise systems to explore alignment and early-stage involvement through confidential discussion.

CCS Guard for Compliance
Regulatory Compliance. Automated. Continuous. Audit-Ready.
CCS Guard is an enterprise software platform transforming how sterile pharmaceutical manufacturers meet Annex 1 contamination control requirements through real-time, automated compliance intelligence.
Eng. Anas Kahwajy Co-Founder of CSS Guard
Ensuring Compliance with Confidence
Your Partner in Contamination Control
At CCS Guard, we understand the complexities of maintaining compliance in sterile pharmaceutical manufacturing. Our innovative solutions support leaders like you in achieving the highest standards of contamination control by offering global monitoring in real time. Together, we can navigate the challenges of regulatory requirements and ensure your operations run smoothly and efficiently.
Professor Osama Abdulrahman Co-Founder of CSS Guard
